I.4 can still be useful if the control sample contains A and B in equal numbers. I.4 is generally unsuitable if samples are unpaired: the ratio of A and B in a sample can be of immediate interest, and any reference to a control sample can be inconvenient or even meaningless. 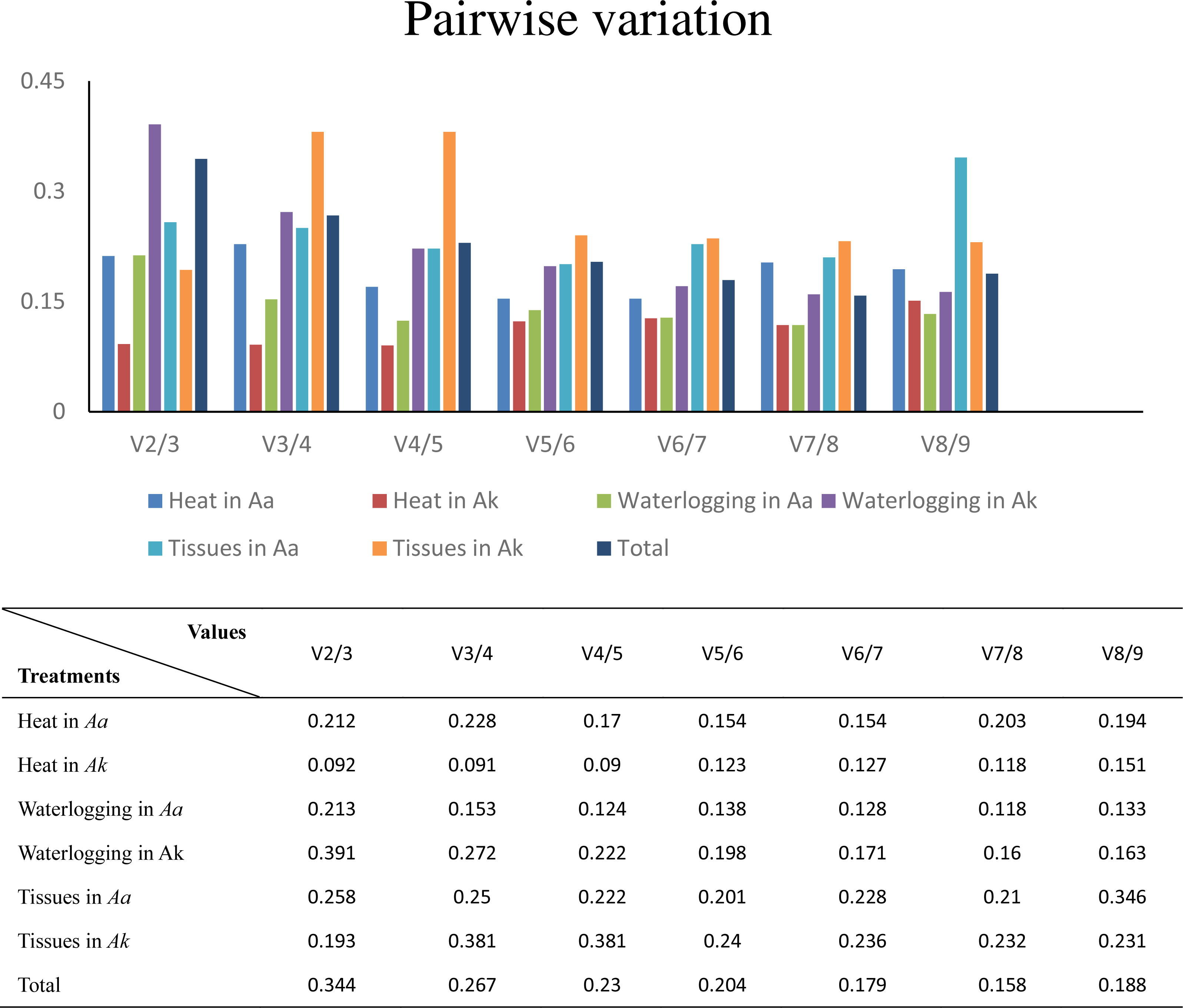
This may not be problematic as such, provided that the ratio of A and B in the interest-sample is of interest only relative to their ratio in a control-sample ( cs): (I.3)The “double-ratio” above can be simplified, eliminating the N Cq,A/ N Cq,B-term, if both N Cq and E are preserved for each target between the samples is and cs that is: ( N Cq) A,is/( N Cq) B,is = ( N Cq) A,cs/( N Cq) B,cs and E A,is = E A,cs, E B,is = E B,cs): (I.4)Although appropriate for paired samples, Eq. The problem lies in the N Cq,A/ N Cq,B-term in conventional settings, the value of this term is unknown. In this paper, we focus on two of these.Ī general limitation is that Eq.I.2 cannot be solved trivially offhand. ĭespite the broad applicability of the technology, several methodological limitations have yet to be addressed. In mainstream qPCR, E is either assumed to have a value of 1 (the “comparative Ct method” or “2 ΔΔ Cq”, or estimated target-specifically from a standard curve (SC). The threshold can be set by various approaches for example, second-derivatives-maximum method, manually setting, and so on. N Cq and the corresponding C q are indirectly defined in the setting of a fluorescence-intensity threshold value. Various technologies exist (reviewed in, ), but the common principle of Real-Time PCR is to obtain fluorescence emissions of an intensity proportional to the amount of target at a given point of time. Indirect measures of N Cqs are estimated by fluorescence sampling. Applying the above relationship, the ratio before PCR of sequences A and B in a given interest-sample ( is) can thus be estimated as: (I.2)To solve the equation above, N Cqs, C qs and Es must be accounted for. The designation “relative quantification” refers to the fact that the amount of the target sequence is estimated relative to that of another (or several ). Usually, E is assumed constant until the onset of PCR exhaustion. The basis of these calculations is the classical PCR equation: (I.1) N 0 is the amount of the target sequence before PCR, N Cq is the amount of target after C q-rounds of PCR, and E is the efficiency of the PCR-amplification. Subsequently, the original amount of the target sequence is calculated from the parameters of the PCR. In essence, an undetectably low amount of a specific nucleic acid target sequence is expanded by PCR to a measurable level. Real-time relative quantitative polymerase chain reaction (qPCR) has long been a favoured principle for relative quantifications of nucleic acid sequences. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
